Metal pickling by inorganic acids 4 – phosphoric acid “H3PO4”
I would like to give a brief overview of those inorganic acids, which are traditionally used in pickling applications.
Phosphoric acid:
Phosphoric acid with a molar mass of 98.0 g/mol and a density of 1.88 t / m³ is in pure, anhydrous state a colorless to white, slightly deliquescent mass. Phosphoric acid is completely miscible with water and the mixture is a colorless liquid. The commercial available concentrations of H3PO4 are in the range of 75 to 90% (m/m).
Table below shows the density / concentration context of phosphoric acid. The numbers were taken from the book Beizen von Metallen, Dr. Ralf Rituper, Eugen G. Leuze Verlag Appendix 6a.
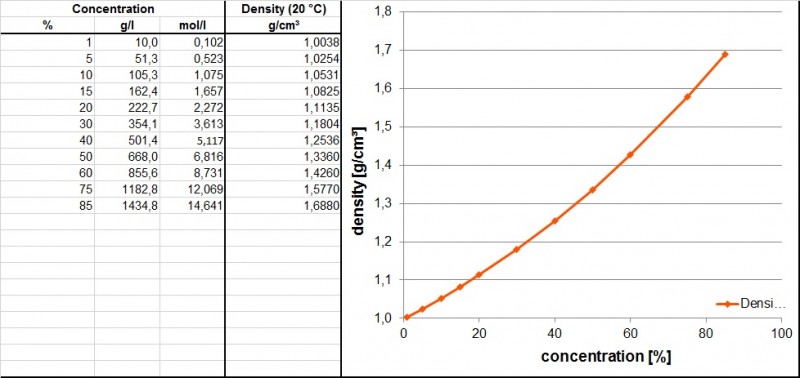
The ortho phosphoric acid is a trivalent, moderately strong acid which dissociates according to the mentioned reaction equations.
H3PO4 + H2O -> H2PO4- + H3O+
H2PO4- + H2O -> HPO42- + H3O+
HPO42- + H2O -> PO43- + H3O+
The dissociation process forms primary, secondary and tertiary phosphates. Undiluted phosphoric acid is present at a pH value of about 0.5 as undissociated H3PO4.
Aqueous phosphoric acid solutions are highly acidic. This turns from primary phosphate solutions through secondary to tertiary phosphate solutions which strongly alkaline.
H3PO4 acts as an aqueous solution practically non-oxidizing. However, a concentrated solution affects all metals and their oxides, above 400 ° C even precious metals like gold and platinum.
The table below shows applications which used phosphoric acid as pickling agent.
The main application is of course in the field of steel pickling. By the use of H3PO4 work pieces are less rusting after treatment and the phosphate substrate represents a good base for subsequent painting.
Phosphoric acid attacks compared to other mineral acids, the base material less. As result it is often used as a component of cleaning products (cleaning, degreasing, rust protection). The solutions also contain other ingredients.

data source: Beizen von Metallen, Dr. Ralf Rituper, Eugen G. Leuze Verlag
See also the blog posts on other inorganic acids: hydrochloric acid; sulfuric acid; nitric acid; hydrofuoric acid

Leave a Reply