Metal pickling by inorganic acids 3 – nitric acid “HNO3”
I would like to give a brief overview of those inorganic acids, which are traditionally used in pickling applications.
Nitric acid:
Nitric acid has a molar mass of 63.0 g/mol and a density of 1.522 t / m³. In the pure state it is a colorless, highly acidic, corrosive liquid which is completely miscible with water. The water HNO3 mixture has azeotropic behavior at a HNO3 concentration of 68.3%(m/m).
The commercial acid concentrations are in the range of 50 to 65 %(m/m), which corresponds to 655-904 g HNO3/liter. The density is also an important indication of the acid concentration and the mentioned range is equal to a density of 1.31 to 1.39 t/m³

The table is showing the relation between density and concentration of nitric acid. The above mentioned, figures were taken from the PERRY’S CHEMICAL ENGINEERS HANDBOOK SEVENTH EDITION.
Nitric acid decomposes at room temperature according to the following equation to nitric oxide, water and oxygen, resulting in a yellow to red coloring. Also the production of red-brown fumes which are emitted is possible. Light led also to the decomposition of nitric acid.
2HNO3 -> H2O + 2 NO2 + 0,5 O2
Chemically, nitric acid is characterized by two important properties. It is one of the strongest inorganic acids and also a strong oxidizing agent. Which property dominates is strongly concentration dependent. In diluted form nitric acid is practically completely dissociated.
HNO3 + H2O -> H3O+ + NO3-
Copper and other noble metals are dissolved by nitric acid. In dilute acid to NO is formed by the solution reaction and in concentrated acid NO2 is produced. Platinum and gold are not solved in nitric acid, and in the case of the silver the formation of silver nitrate AgNO3 take place.
Another interesting property of nitric acid is, for example that iron, aluminum and chromium are dissolved by dilute acid. In concentrated acid, the listed metals forms a dense layer and a passivating effect stops the dissolution process.
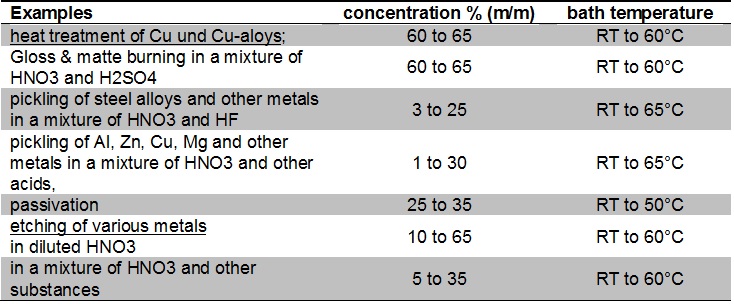
Data source: Beizen von Metallen, Dr. Ralf Rituper, Eugen G. Leuze Verlag
See also the blog posts on other inorganic acids: hydrochloric acid; sulfuric acid; phosphoric acid; hydrofuoric acid

Leave a Reply